
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Include the color of the balloon used to model each electron domain. For each of these electron geometries: Sketch the shape of the VSEPR geometry formed. you can visualize each electron domain geometry. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion The flow chart below gives a schematic view for how one can figure out the molecular geometry from the molecular formula.Students and scientists can use these charts to create three-dimensional diagrams that represent molecules. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. 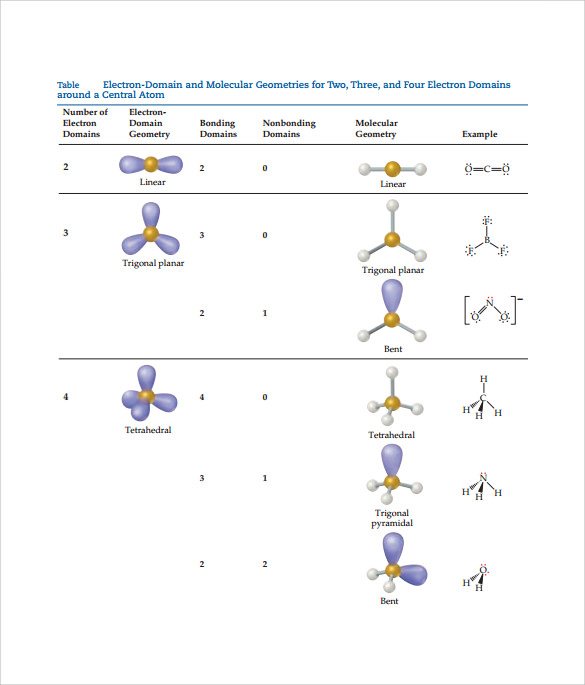
The second figure serves as a visual aid for the table. The table of molecular geometries can be found in the first figure. A table of geometries using the VSEPR theory can facilitate drawing and understanding molecules. The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral. Molecular geometry is the science of representing molecules in a three-dimensional manner. Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the VSEPR theory.Molecular geometries take into account the number of atoms and the number of lone pair electrons. Once you know PCl 5 has five electron pairs, you can identify it on a VSEPR chart as a molecule with a trigonal bipyramidal molecular geometry.Molecular Geometry: The number of bonding electron pairs should be calculated to find the molecular geometry. Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Electron Geometry: The number of total electron pairs should be calculated to find the electron geometry. There are five electron pair geometries: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
